Spring
Spring
… and suddenly it was spring!
Enjoy a picture …
What? Not “astronomy” enough for you? Astronomy is the REASON we have cherry blossoms in spring!
… and Three Carnivals!
Carnival #142 – with wonderful illustrations, and featuring two of my posts! Check it out.
![]()
~ A l i c e !
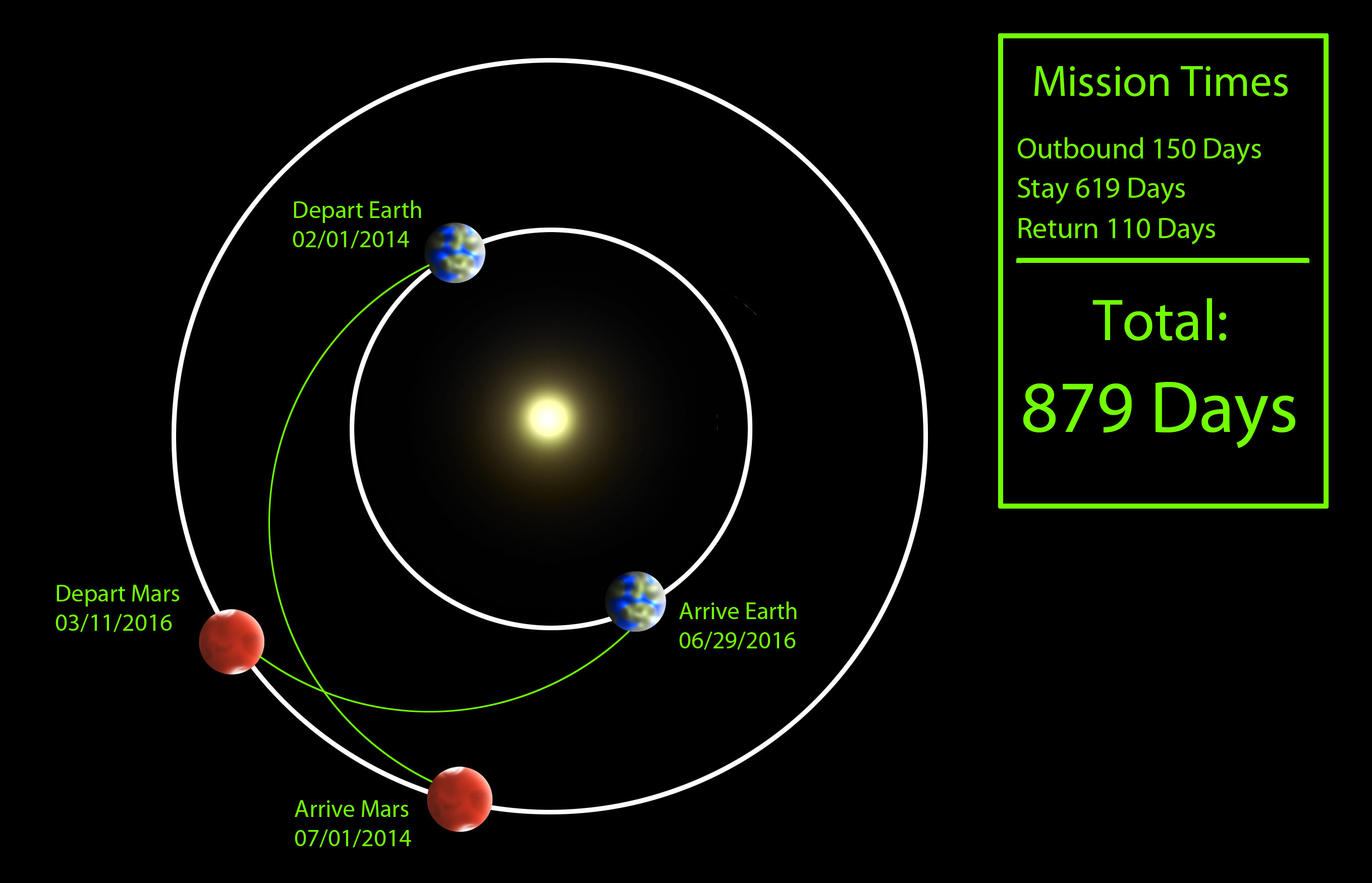
 Which Way to Mars?
Which Way to Mars?
How long does it take to get to Mars? Well, that depends. As per usual I’ll give you the short answer first: 5-7-ish months.
Depending On …
- Energy
- The faster you go, the more energy you must expend to get there. Energy is expensive.
- When You Leave
- You could leave any time you wanted to, but if you don’t leave at the right time you’ll just end up chasing after Mars, or doodling around the orbit waiting for Mars to catch back up to you. It’s like catching a bus – you don’t want to wait around at the bus stop too long, but you also don’t want to miss the bus.
- Length of Stay
- If you’re going to all the trouble of going to Mars, I would think you’d want to spend a little time there. Then, your return journey has to be timed just right
Some Choices
A Hohmann transfer orbit is one of the most common ways to move between orbits. It is energy-efficient without being incredibly slow.
The Classic Choice:
So, if you do a couple of plain Hohmann transfer orbits – one on your way to Mars, one on your way back, you end up with about 7.5 months each direction with a nice long stay of over a year on Mars.
Speed It Up:
Well, why waste all that time en route? If you up your energy consumption you can move between orbits a little faster. This is very similar to a Hohmann transfer orbit, but significantly quicker. Travel time is more like 5 months each direction, and you still get to spend almost two years on Mars.
Early-Return
What if you get there and need to come back? What if you’re just not sure what might happen and don’t want to invest a lot in spending time there? If your goal is more to prove that it is possible to go there than to spend time there, you might prefer an early-return mission. You sacrifice a lot on the return mission: it takes almost 10 months to get home. For a “footprints on Mars” mission (similar to our first Moon missions) this gets you just under a month of study-time on Mars. Also, if things start to go wrong early on in a longer mission, you could abandon the remaining mission and return early using this path. Plus you might get to swing by Venus!
Ultra-Slow
There are two more quick options I’d like to mention. The ultra-slow route – spiraling out from Earth’s orbit, eventually reaching the orbit of Mars. The Dawn spacecraft is using an orbit like this and a low-energy ion engine to reach the asteroid belt over the course of six years.
Don’t Come Back
It sure takes months off your travel time if you just go and don’t return. Like the settlers of the American West and all over the world – the people who make a new world their own are the people who set out with that in mind. This is a much cheaper mission – and there are some fascinating arguments for it.
Want More?
http://nssdc.gsfc.nasa.gov/planetary/mars/marsprof.html
http://image.gsfc.nasa.gov/poetry/venus/q2811.html
http://dawn.jpl.nasa.gov/mission/trajectory.asp
![]()
~ A l i c e !
 Feed Trouble
Feed Trouble
I’m having trouble filtering my RSS feed correctly. Please bear with me, and if you’re not taking my Astr 100 class this term you’re welcome to ignore any posts with the date as the title that may have accidentally shown up in your feed reader.
It will be fixed soon!
![]()
~ A l i c e !
P.S. Take advantage of our beautiful sky and go look for Mars, Vesta, and ISS among the stars over Seattle this weekend. Mars and Vesta should be visible anywhere in the northern hemisphere, and you’ll need binoculars for Vesta unless you live somewhere with a lot less light than Seattle’s got.
 Communicating With Mars
Communicating With Mars
One of the most basic questions when it comes to Martian expeditions is communication, namely “how long does it take to communicate with Mars?” The simple short answer is 4-20 minutes, depending on where the Earth and Mars are in their orbits. It also gets more difficult when Mars is near or behind the Sun from our point of view.
Interplanetary Internet
You’re probably quite used to looking up the answers to your questions on the internet anytime you want, but with the delay in communication in space, the internet can’t work quite the same way – besides the fact that you’ll be using a connection that feels like a very, very slow modem. The way webpages are designed now that would take forever.
Data Rates
Of the missions currently at the red planet, the Mars Reconnaissance Orbiter (MRO) has the ability to transmit the most data. Its data speed to and from Mars is about 6 megabits per second (about twice as fast as the connection I’m currently on), but that’s not continuous – it is only when MRO can “see” Earth. Since Mars is rotating, the rovers are periodically out of contact, and the satellites lose their connection when they orbit to the far side of the planet.
Eeep! We’re using speed in two different ways. Don’t confuse the speed at which the data is transmitted (the amount of data per second) with how long it takes the data to get to Earth. MRO may transmit at 6 megabits per second, whereas Spirit can only transmit directly to Earth at a maximum of 12,000 bits per second (that’s 0.01 megabits per second – like using a very old dial-up modem!), but still both messages will take the exact same amount of time to get to Earth: between 4 and 20 minutes, depending on the time of year.
Relays
Most messages from space missions go through relay satellites, for example the Mars Rovers often send data through MRO. This in itself might make interplanetary internet a reality, if we can overcome some of the blocks – like the delay causing excessive timeouts among many others. There are some folks studying this problem, and there are even proposed solutions.
Incidentally, the International Space Station just got live internet for the first time – really it’s a connection to a computer in Mission Control – and that computer is connected to the internet. That limits the information that really needs to be transmitted to the Space Station.
Would You Like to Receive a Message from Mars?
Would you like to get some tourist snapshots of Mars? The surface of Mars? Well, your chance to point the HiRISE camera at your favorite part of Mars is here.
More Info:
http://deepspace.jpl.nasa.gov/dsn/
http://marsrovers.nasa.gov/mission/comm_data.html
![]()
~ A l i c e !
 Spirit at Troy
Spirit at Troy
Happy Valentine’s Day and Happy Lunar New Year, Spirit! Here are some of our imaginings about how you look as a station!
Advanced Entries
There are six secrets hidden in this image. Emmanuel says: “Some are Mars exploration related. Kind of. Can you find them?” (I’ve found one so far … he sent me the answers, but I haven’t opened that file yet.) If you want the answers, comment below and I’ll e-mail them to you.
Also of note – as this is pixel-art, the display, zoom, and rendering are important. See below* for extensive detail, or visit PixelJoint.com to increase your exposure to the medium. The original size of Silence at Troy is available here.
This entry is from Gary Kezele, accomplished artist and astronomy-visualizer. Bear with us while we get a better scan of this image, but for now you can see a decent version of Gary’s submission! Please click to see it in more detail.
Both these entries show amazing attention to detail – and you can tell they looked closely at the photos of Spirit’s location.
Hobby Entries
From @MamaJoules come two model entries – one from each son. I love them both, and bet that it was a lot of fun to build rovers out of Legos and then try to position them just right in the “sand.” :)
The camera mast is great, there are lots of fun details, and the flag is a nice touch too!
Once again, the camera mast is in evidence, and the rover is just like Spirit: it has six wheels (I wonder if all of them work on this model).
Thanks to everyone who submitted – I’ll be sending you all some fun NASA postcards. :) I’ll also pass these along to as much of the Spirit team as I can.
More
These were not submissions, and you’ve seen them before, but I had to include them in this post because I thought you’d enjoy them.
Go see more of Stuart’s Mars Art.
*Pixel Art
Emmanuel has sent these details about viewing pixel art, which I found fascinating.
Pixel-art makes for small images and files. They should ideally be viewed at 200%, 300% max. You should be able to just see the pixels without them being overwhelming.
Displaying pieces at 100% makes them look very small, and the pixels are lost on good monitors.Using the browser zoom is not an option! All current browsers use ‘bilinear filtering’ for zooming images (and most image viewing software too), the result is horribly blurry on pixel-art (you might have noticed this if you browsed the Pixeljoint gallery and used the zoom function. However, from Firefox 3.6, websites can opt for an old-fashioned unfiltered zoom function. This is not something the user can do, it’s only a website-side option. So if you visited Pixeljoint using Fx 3.6, you didn’t notice any problem.
You should note, I did not do anything website-side to enable this function in Firefox. All his images are displayed exactly at the resolution I got from him though. Clicking the main image takes you to his 300% zoom version.
Want More?
The Mars Exploration Program at JPL
Thanks all! Let’s do it again sometime. Thanks to the NASA Solar System Ambassadors Program for the support.
![]()
~ A l i c e !
 Why Is Rust Red?
Why Is Rust Red?
This post is at a deeper level of understanding than a lot of what I write. If you’re lost by step one or two – review spectra and come back!
The Premise
When I think of a good question, I just can’t let it rest. Here’s what I tweeted today:
Why does iron make things red? Mars – blood – iron tablets – hematite scratch tests? (I don’t know)
I’ll clarify/deepen my question: Assume your audience groks spectra and color (in depth). Now answer “Why does iron oxide make things red?”
People started sending me parts of the answer, and I just had to figure it out.
The Puzzle Pieces
Together these pieces make up an answer.
- All the colors that are not absorbed are the colors you see.
- Spectra for single atoms tend to be simple – electron energy levels are fairly well defined so one detects discrete absorption lines.
- Spectra for molecules (such as the various iron oxides) are much more complex, because the electron energy levels are less well defined. In fact there are significantly more energy states and vibration possibilities for molecules, so those discrete lines end up as whole bands of absorption.
- Iron oxide happens to be a molecule that absorbs energy in pretty much every state corresponding to the purple, blue, green, and into the yellows – leaving the reds reflected.
- Blood is a little different, but related. Hemoglobin has hemes – those hemes carry the Iron in the middle. When that heme interacts with O2 (note I did NOT say bonds to) the whole shape of the molecule changes causing the orbitals to move, which changes the energies that can be absorbed by the electrons. These shift towards those blues again, leaving (once again) the reds reflected.
- Oh, and hematite is grey/silver, but the scratch is reddish because in the hematite crystal structure the orbitals are stuck in one state, but once you disrupt that structure – by grinding it up – the orbital positions can change allowing different amounts of energy to be absorbed – like the blues, leaving the reds. Again.
Whew.
Thanks @superacid & Doug
Sources:
http://www.newton.dep.anl.gov/askasci/bio99/bio99423.htm
Doug McGregor
And I used Wikipedia to get some definitions – like “heme” and “ligand.” That doesn’t mean you should cite Wikipedia as a trustworthy source though!
I am accepting corrections and clarifications to this post – though I’m not going to go into the basics of spectra here. Maybe another time?
![]()
~ A l i c e !
 The Rocket and the Sundog
The Rocket and the Sundog
Watch this video from just a little earlier today: the SDO rocket’s shockwave distrupts a sundog!
 Mineral Indications of Water
Mineral Indications of Water
What you need to know:
Carbonates (like antacids that you dissolve in water) dissolve easily in water, and are deposited easily out of water. Sulfates (which are why Yellowstone’s fantastic mud-pots smell awful) are similar. Silica-rich deposits are more commonplace – so you need more detail to tell if they’re from water or not.
Up until now you’ve taken it for granted when you read in an article that a mineral discovered on Mars indicates the prehistoric existence of water. But how can a certain mineral in a rock tell you that there was once water? How can it tell you if that water was more recent or more ancient?
Carbonates
There are many carbonates, but calcium carbonate is probably the most familiar to you – and one of the most common carbonates. As you know from taking Tums or Alka-Seltzer – these dissolve easily in water. Many seashells are made of carbonates as well, and when those animals die, the shells are dissolved into the ocean and eventually that calcium carbonate is redeposited as limestone. Like that, all carbonates are usually formed in “hydro” and hydrothermal situations, and are also later dissolved by even a slightly acidic situation. Although this example only represents redeposition of carbonates – they are also deposited primarily (in the first place) by water as well. Finally, in order for carbonates to last a geologically long time they must be protected from water and other acids. Carbonates are either a good indication of water activity in the geologically recent past, or there hasn’t been any water near them in a long, long time.
Carbonates in ALH84001 (one of the meteorites from Mars)
Sulfates
Sulfate minerals are similar to carbonates in terms of their indication of water – they are also water-soluble, and occur in similar places to carbonates: embedded with limestones, etc. Sulfates tend to need more “thermal” in the term “hydrothermal” than carbonates – which is why you find a lot in volcanic regions like Yellowstone. That rotten-egg smell associated with hydrothermal areas on earth is due to the sulfur in sulfates. Sulfates are a great indicator for water activity paired with heat.
Spirit Unearths Sulfates on Mars
Silica
Almost every rock on Earth is silica-based. The presence of silicates in and of themselves is not an indication of water, current or prehistoric. More analysis or specificity is needed. After oxygen (yes, oxygen!) silica is the most common element in the Earth’s crust, as well as the crust of the Moon, and what we know of the surface of every other terrestrial planet.
Where’d I Get My Info?
Klein, Cornelius. The 22nd Edition of the Manual of Mineral Science.
![]()
~ A l i c e !
 Carnival of Space #139
Carnival of Space #139
Welcome Mama Joules to the astroblogger community – stop by Carnival of Space #139 hosted by her!
![]()
~ A l i c e !